Ethylene Oxide ETO Sterilization
Ethylene oxide (EO or EtO) is a widely utilized low-temperature gaseous sterilization method for various healthcare products, particularly single-use medical devices. This process involves using a vacuum to efficiently permeate the surfaces of most medical devices, making it suitable for a wide range of materials.
Ethylene oxide gas is used to sterilize medical devices due to its effectiveness and compatibility with various materials.
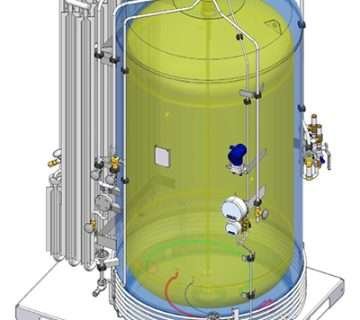
This colourless gas, while highly effective, possesses flammable and explosive properties. The process of ETO sterilization involves four critical parameters, namely:
Gas Concentration: The operational range of gas concentration typically falls between 450 to 1200 mg/l. This concentration is crucial to ensure optimal sterilization efficacy.
Temperature: The temperature range during the ETO sterilization process is typically maintained between 37 °C and 63°C. This specific temperature range is selected to achieve effective sterilization without damaging the medical devices.
Relative Humidity: Maintaining a relative humidity range of 40 to 80% is essential during ETO sterilization. The presence of water molecules helps facilitate the transportation of ETO to reactive sites, enhancing the sterilization process.
Exposure Time: The recommended exposure time for ETO sterilization typically ranges between 1 to 6 hours. This duration ensures sufficient contact between the gas and the medical devices, enabling thorough sterilization.

Ethylene oxide sterilization, among all available alternative sterilization methods, generates the highest level of concern. Whether utilized alone or in combination with CFCs, EtO sterilization poses environmental and bodily harm. However, with adequate attention to establishing and enforcing standards that regulate EtO sterilization, stakeholders can reasonably ensure its viability for the coming years.

Ethylene Oxide Uses
Manufacturers primarily employ ethylene oxide to manufacture other chemicals for consumer and industrial use. One main chemical created from EO is ethylene glycol, which is used widely across products from brake fluid to industrial solvents.
EO is also used to produce healthcare products and sterilize medical devices.
Ethylene oxide uses include:
EtO Sterilization
Although ethylene oxide sterilization (EtO) is commonly used in the manufacture of sterile medical devices, concern about residues of the sterilant in the sterilized products has led to an increase in the use of alternative methods;
Applications
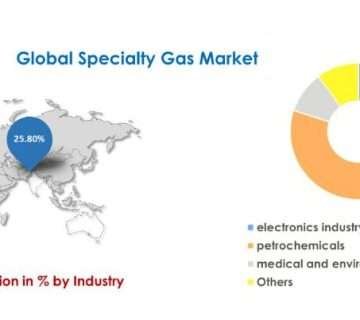
ETO (ethylene oxide) sterilization has a wide range of applications across various industries. Some of the notable applications of ETO sterilization include:
Healthcare and Medical Devices: ETO sterilization is extensively used in the healthcare sector for sterilizing a diverse range of medical devices, including surgical instruments, implantable devices, catheters, syringes, and single-use medical products. It effectively eliminates microorganisms and ensures the sterility of these critical healthcare items.
Pharmaceuticals: ETO sterilization is employed in the pharmaceutical industry to sterilize drug products, particularly those that are heat-sensitive or cannot withstand high-temperature sterilization methods. It helps maintain the integrity and efficacy of pharmaceutical products while ensuring their sterility.
Food and Beverages: ETO sterilization finds application in the food and beverage industry for decontaminating spices, herbs, and other food ingredients susceptible to microbial contamination. It helps extend the shelf life of these products while ensuring they are safe for consumption.
Laboratory and Research Equipment: ETO sterilization is utilized in laboratories and research facilities to sterilize equipment, glassware, and supplies. This ensures a sterile environment for conducting experiments and maintaining the integrity of samples.
Cosmetics and Personal Care Products: ETO sterilization is employed in the cosmetics and personal care industry to sterilize various products, such as creams, lotions, powders, and other beauty or personal care items. It helps eliminate microorganisms and ensures product safety.
Textiles: ETO sterilization is used in the textile industry to sterilize fabrics and garments, particularly those used in healthcare settings or for sensitive individuals. It helps eliminate bacteria, viruses, and other pathogens that may be present on the textile surfaces.
Packaging Materials: ETO sterilization is applied to sterilize packaging materials like pouches, trays, and bottles used for medical devices, pharmaceuticals, and other sensitive products. It ensures the sterility of the packaging, preventing contamination during storage and transportation.
These are just a few examples of the diverse applications of ETO sterilization. Its ability to penetrate various materials and effectively eliminate microorganisms makes it a valuable sterilization method in industries where other methods may be impractical or ineffective.
Ethylene oxide (EtO) sterilization is a widely used method in various industries, including healthcare, pharmaceuticals, and food processing. However, concerns have been raised regarding the environmental and health impacts associated with EtO residuals. In response, researchers and scientists have delved into exploring strategies to mitigate these concerns, aiming to reduce EtO residuals while maintaining the required level of sterility.








No comment